what does it mean for a moecule to have greater polarizabililty
Polarizability
- Folio ID
- 1663
Polarizability allows us to better understand the interactions betwixt nonpolar atoms and molecules and other electrically charged species, such as ions or polar molecules with dipole moments.
Introduction
Neutral nonpolar species have spherically symmetric arrangements of electrons in their electron clouds. When in the presence of an electric field, their electron clouds can be distorted (Effigy \(\PageIndex{1}\)). The ease of this baloney is defined as the polarizability of the cantlet or molecule. The created distortion of the electron cloud causes the originally nonpolar molecule or atom to larn a dipole moment. This induced dipole moment is related to the polarizability of the molecule or atom and the strength of the electrical field by the following equation:
\[μ_{ind} = \blastoff E \label{1}\]
where \(Due east\) denotes the strength of the electric field and \(\blastoff\) is the polarizability of the atom or molecule with units of C kii5-1.
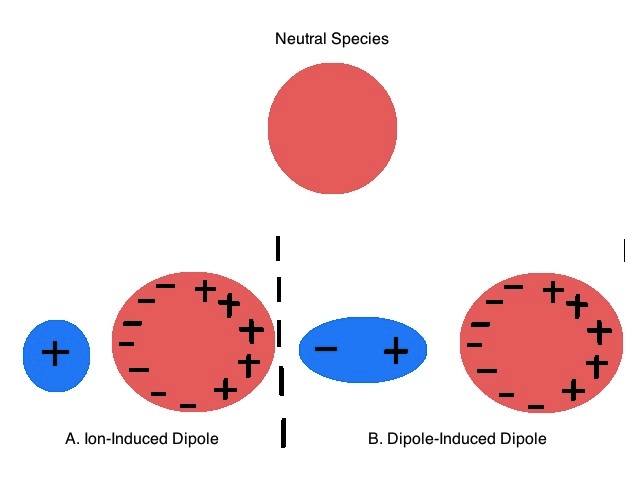
In general, polarizability correlates with the interaction between electrons and the nucleus. The amount of electrons in a molecule affects how tight the nuclear charge can control the overall charge distribution. Atoms with fewer electrons will have smaller, denser electron clouds, as at that place is a strong interaction between the few electrons in the atoms' orbitals and the positively charged nucleus. There is besides less shielding in atoms with fewer electrons contributing to the stronger interaction of the outer electrons and the nucleus. With the electrons held tightly in place in these smaller atoms, these atoms are typically not easily polarized past external electric fields. In contrast, big atoms with many electrons, such as negative ions with backlog electrons, are hands polarized. These atoms typically have very diffuse electron clouds and large diminutive radii that limit the interaction of their external electrons and the nucleus.
Factors that Influence Polarizability
The relationship between polarizability and the factors of electron density, diminutive radii, and molecular orientation is as follows:
- The greater the number of electrons, the less control the nuclear charge has on charge distribution, and thus the increased polarizability of the atom.
- The greater the distance of electrons from nuclear accuse, the less control the nuclear accuse has on the accuse distribution, and thus the increased polarizability of the atom.
- Molecular orientation with respect to an electric field tin affect polarizibility (labeled Orientation-dependent), except for molecules that are: tetrahedral, octahedral or icosahedral (labeled Orientation-independent). This gene is more of import for unsaturated molecules that contain areas of electron-dense regions, such as 2,4-hexadiene. Greatest polarizability in these molecules is achieved when the electrical field is applied parallel to the molecule rather than perpendicular to the molecule.
Polarizability Influences Dispersion Forces
The dispersion force is the weakest intermolecular force. It is an attractive force that arises from surrounding temporary dipole moments in nonpolar molecules or species. These temporary dipole moments arise when in that location are instantaneous deviations in the electron clouds of the nonpolar species. Surrounding molecules are influenced by these temporary dipole moments and a sort of chain reaction results in which subsequent weak, dipole-induced dipole interactions are created. These cumulative dipole- induced dipole interactions create attractive dispersion forces. Dispersion forces are the forces that make nonpolar substances condense to liquids and freeze into solids when the temperature is depression plenty.
Polarizability affects dispersion forces in the following ways:
- Every bit polarizability increases, the dispersion forces also become stronger . Thus, molecules attract one some other more than strongly and melting and humid points of covalent substances increase with larger molecular mass.
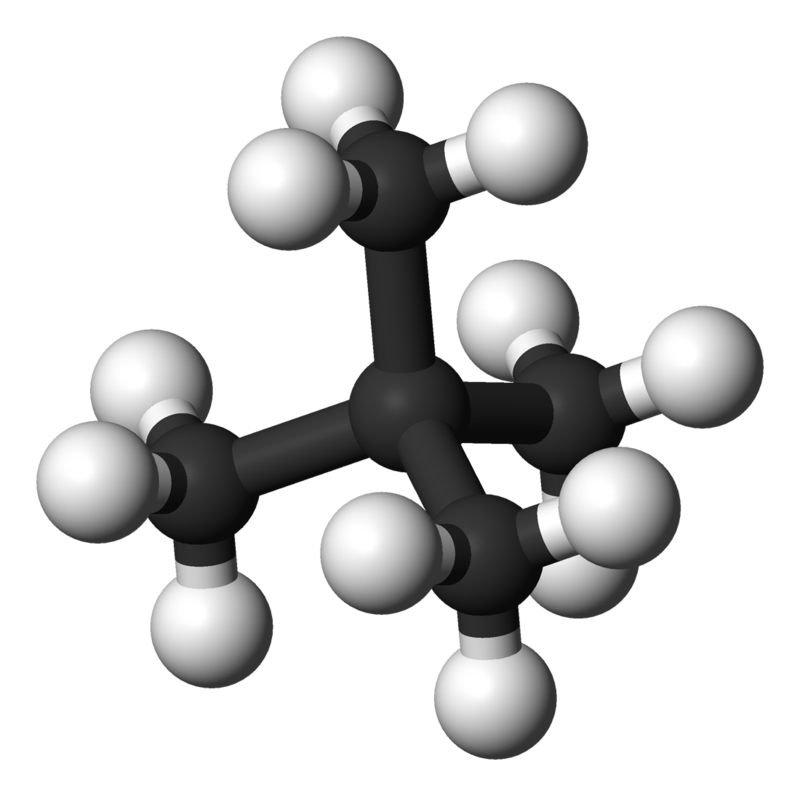
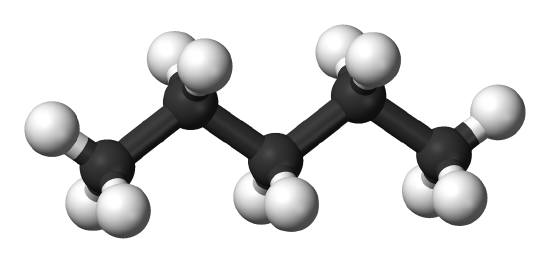
- Polarazibility also affects dispersion forces through the molecular shape of the affected molecules. Elongated molecules accept electrons that are hands moved increasing their polarizability and thus strengthening the dispersion forces, (Effigy \(\PageIndex{2}\)). In dissimilarity, small, compact, symmetrical molecules are less polarizable resulting in weaker dispersion forces.
The relationship between polarizability and dispersion forces can exist seen in the post-obit equation, which can exist used to quantify the interaction between 2 similar nonpolar atoms or molecules:
\[ 5 = \dfrac{-3}{4} \dfrac{\alpha^ii I}{r^half dozen} \label{2}\]
where
- \(r\) is the altitude between the atoms or molecules,
- \(I\) is the offset ionization energy of the atom or molecule, and
- \(\alpha\) is the polarizability constant expressed in units of 10003.
This expression of \(\alpha\) is related to \(\alpha'\) by the post-obit equation:
\[\blastoff' = \dfrac{\blastoff}{4 \pi \epsilon_o} \label{3}\]
To quantify the interaction between unlike atoms or molecules (A and B) the Equation \(\ref{two}\) becomes:
\[ V = \dfrac{-3}{2}\dfrac{I_AI_B}{I_A+I_B} \dfrac{\alpha_A \alpha_B}{r^6} \characterization{4}\]
References
- Petrucci, Ralph H., et al. Full general Chemical science: Principles and Mod Applications.Upper Saddle River, NJ: Prentice Hall, 2007
- Chang, Raymond. "Chapter thirteen: Intermolecular Forces/ Ion-Induced Dipole and Dipole-Induced Dipole Interactions/ Dispersion, or London, Interactions." Concrete Chemical science for the Biosciences. Sansalito, CA: Academy Science, 2005. 495-98. Print.
- Housecroft, Catherine E., and Alan G. Sharpe. "Chapter half-dozen: Structures and Energetics of Metallic and Ionic Solids." Inorganic Chemical science. Harlow [etc.: Prentice Hall, 2008. 173-74. Print.
- Miessler, Gary Fifty., and Donald A. Tarr. "Chapter 6: Acid-Base of operations and Donor-Acceptor Chemistry." Inorganic Chemistry. Upper Saddle River, NJ: Pearson Education, 2004. 183-86. Print
Contributors and Attributions
- Kelly Cox (UCD), Dana Reusser (UCD)
fourniermorelesucity78.blogspot.com
Source: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Physical_Properties_of_Matter/Atomic_and_Molecular_Properties/Intermolecular_Forces/Specific_Interactions/Polarizability
0 Response to "what does it mean for a moecule to have greater polarizabililty"
Post a Comment